Researcher Spotlight
Using high and low energy parts of the spectrum more efficiently - MIT-Harvard Center for Excitonics
Graduate student Priya Jadhav is trying to solve a problem that relies on a singlet exciton and a pair of its “cousins,” a triplet exciton. Priya is currently a fourth year graduate student who, after training in electrical engineering and a professional role in the software industry, decided to pursue doctoral work that would involve her in work positioned in between electrical engineering and physics or, as Priya calls it, in applied physics.

She learned of Marc Baldo’s work and asked to join when she was admitted to MIT. Priya is focused on very tiny particles called excitons and using them in a unique way to extract energy from the high- and low-energy regions of the solar radiation spectrum.
The sun presents both opportunities and barriers to generating electric current efficiently. The sun’s radiation spectrum contains various wavelengths of visible and invisible light. The visible portion lights our journey to work or school each day. For the most part, this is what is currently used by conventional solar cells to convert light to electricity. But the invisible spectrum – especially the infrared – offers us much more total energy “real estate” to work with and thus the challenge is to figure out how to exploit it. The fact is that traditional silicon solar cells, which can be seen in the form of the blue-black flat panels installed on rooftops, actually use only about 25% of the potential solar energy available in the infrared.
Infrared light is transmitted by lower energy photons. The problem is that if you tune a conventional solar cell to catch the infrared, it cannot also efficiently convert visible light into energy. Instead, it would turn most of the visible photons into heat. What is needed is the equivalent of two solar cells – one optimized for the visible and one optimized for the infrared. Such tandem solar cells are currently built, but these can be expensive and are presently impractical with commonly used silicon. Thus, Priya’s challenge is to try to do all of this in one solar cell – take advantage of energy in the infrared and also in the visible part of the spectrum – and by using new materials, the organics.
Researchers in the Solar Antennas group have turned to organic materials for several reasons. They are inexpensive and are five-to-ten-times a better absorber than silicon. Organics are also composed of molecules so a researcher can do many things to alter their state, and test absorption and other attributes needed for a more efficient transfer of light to energy. In addition, organics are easy to form into flexible, very thin substrates or films, enabling versatility in how researchers can examine molecules in a high energy state.
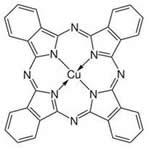
Copper phthalocyanine – every corner in this schematic is a carbon atom even though it isn’t marked. And many of these carbon atoms have a hydrogen atom attached to them.
Now, meet the exciton. An exciton is an excited state formed when light is absorbed by matter such as an organic material. The exciton is an “electron-hole” pair. When excited by light, the electron leaves behind the “hole,” which is like a bubble in an ocean of electrons, and is effectively positively charged. The exciton is formed when the electron and the hole are bound together.
Excitons can be used to mediate the transfer of light into useable electrical energy. Unlike electrons, which must conserve their charge, excitons can split, so this gives the researcher the ability to convert high energy photons into infrared photons without wasting the difference as heat. During Priya’s experiment, the aim is to incite an exciton to split (or undergo fission) within a solar cell. In an intricate experiment, her group created a very thin coating on a solar cell. The exciton is successfully split upon absorption by tetracene, and its “offspring,” the triplets, start to diffuse through the first layer and into the solar cell formed by the second and third layers.[i] This approach doubles the number of charges extracted from the high energy photons without the necessity of an expensive tandem cell!
[i] P. Jadhav, A. Mohanty, J. Sussman, J. Lee, M. Baldo,“Singlet Exciton Fission in Nanostructured Organic Solar Cells,” NanoLetters 2011, 11, 1495-1498.






